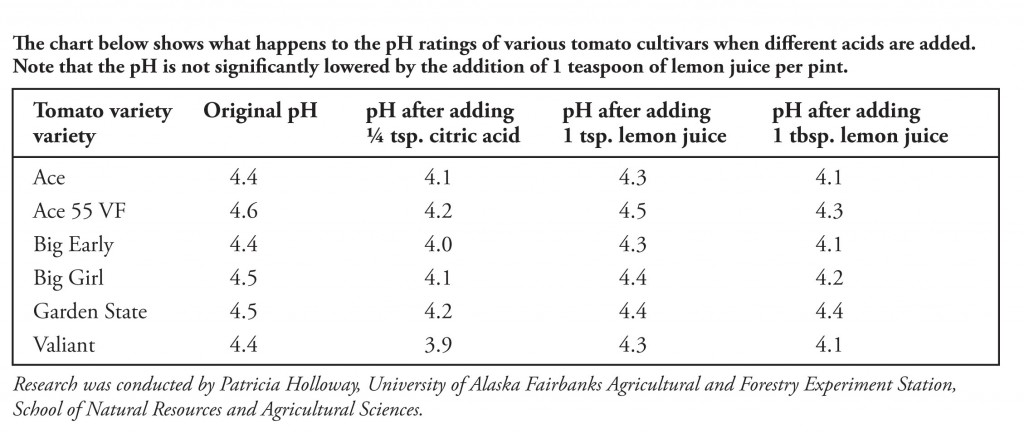
SOLVED: Lemon juice has a pH of about 2.5. Assuming that the acidity of lemon juice is due solely to citric acid, that citric acid is a monoprotic acid, and that the

Experiment for the Determination of amount of citric acid in lemon juice | Acid base titration - YouTube

The Case for Bottled Lemon Juice in Canning • AnswerLine • Iowa State University Extension and Outreach

PDF) Quantitative Assessment of Citric Acid in Lemon Juice, Lime Juice, and Commercially-Available Fruit Juice Products

SOLVED: Lemon juice has a pH of about 2.5. Assuming that the acidity of lemon juice is due solely to citric acid, that citric acid is a monoprotic acid, and that the

Lemon juice has a pH = 2.1 . If all the acid in lemon is citric acid (H Cit. hArr H^(+)+Cit^(-1)... - YouTube