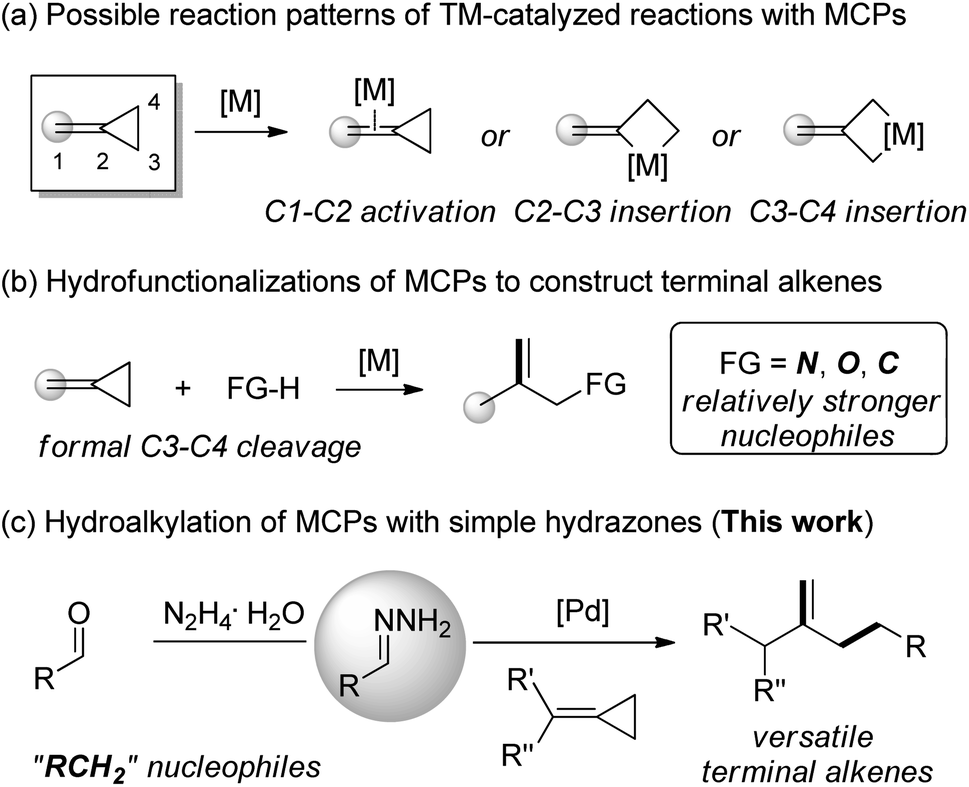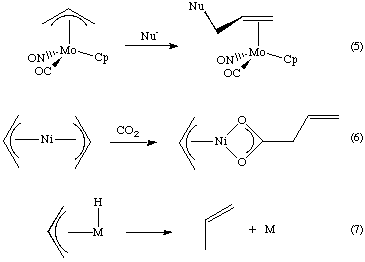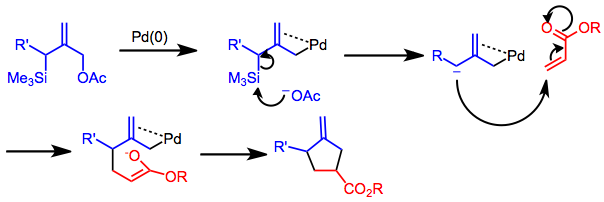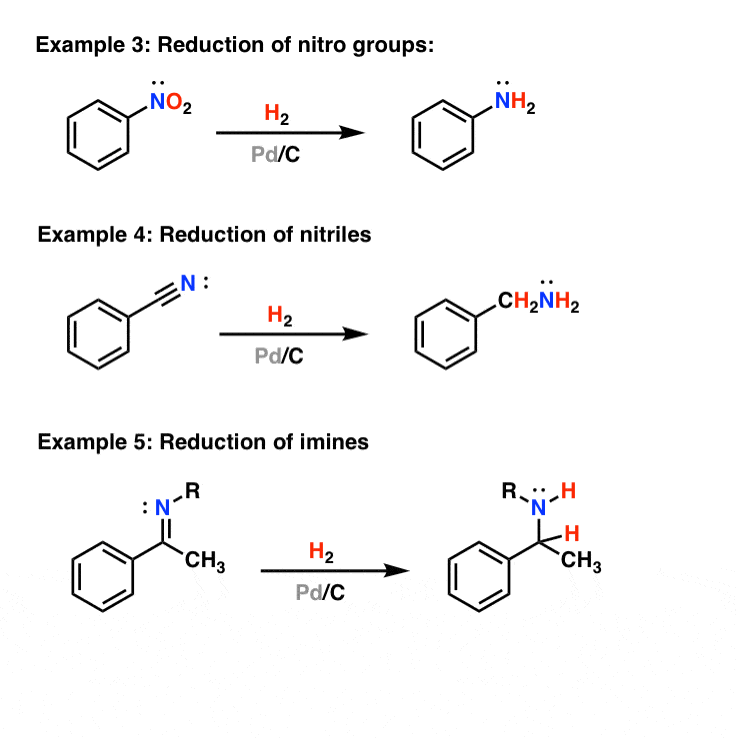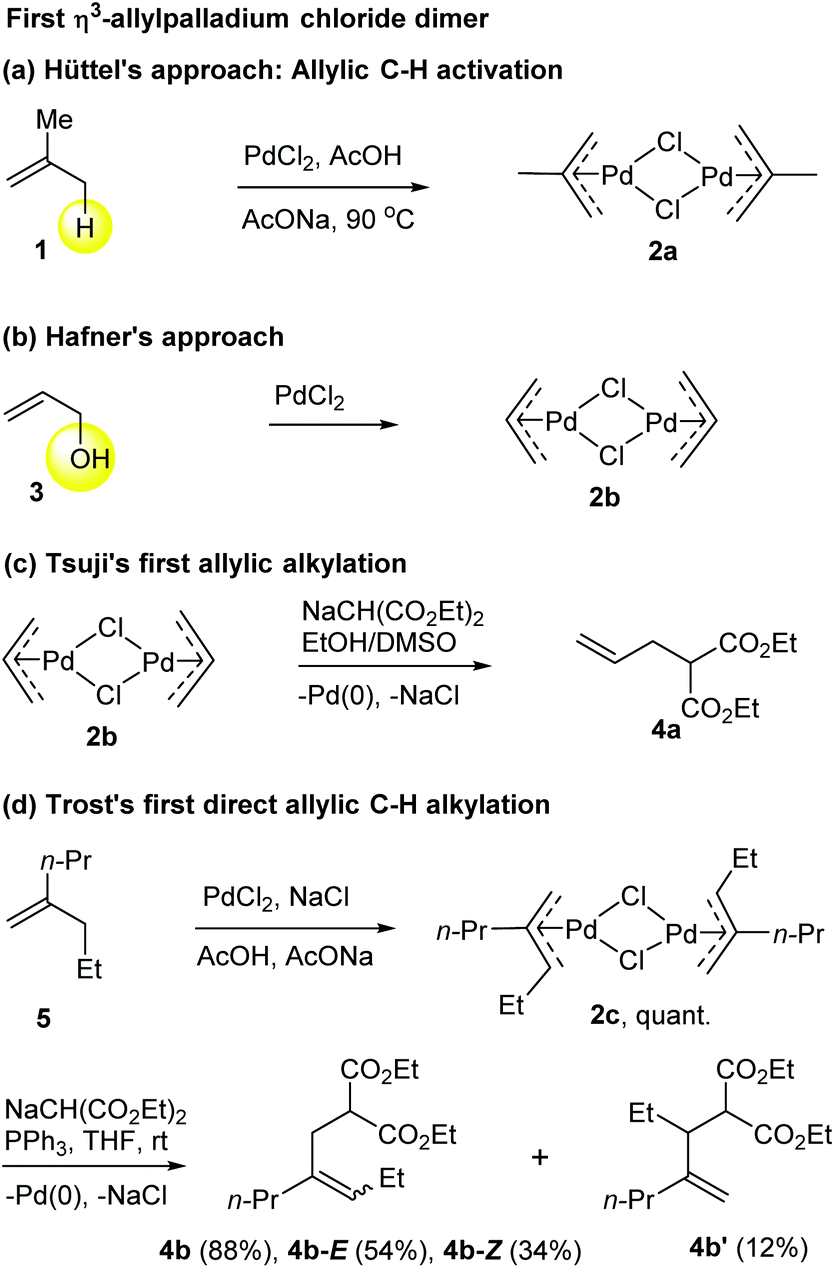
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01725A

Palladium‐Catalyzed Oxidation Reactions of Alkenes with Green Oxidants - Hu - 2019 - ChemSusChem - Wiley Online Library
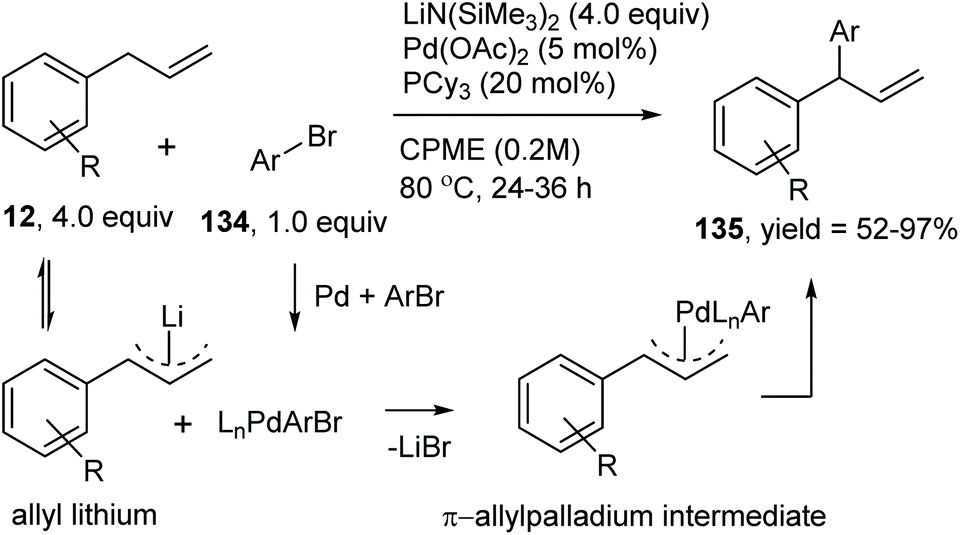
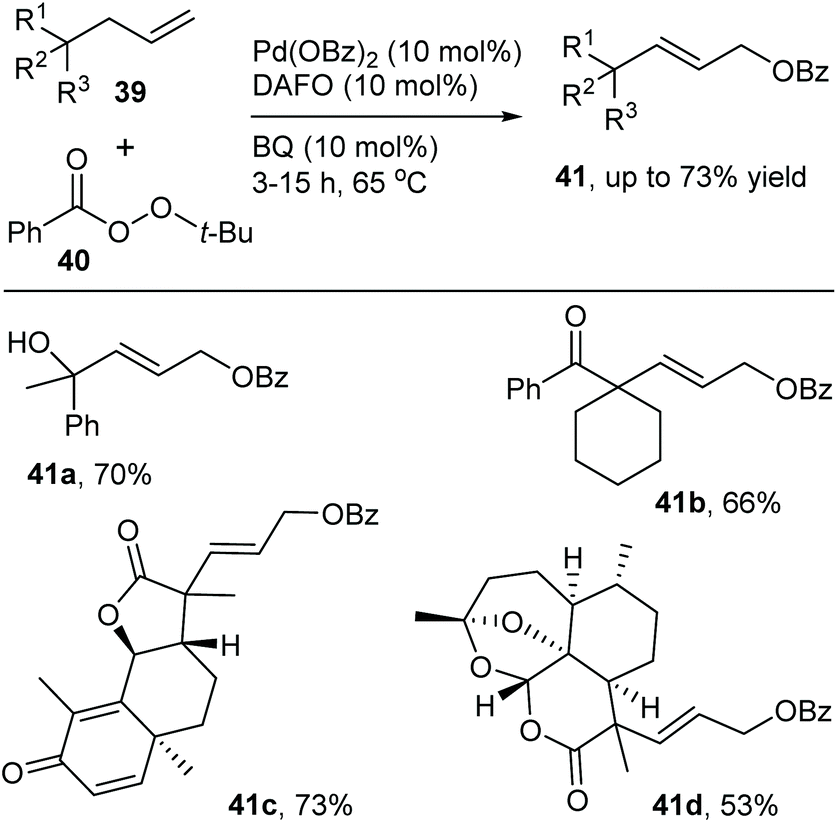
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01725A

Synthesis, characterization, and reactivity of (π-allyl)palladium(II) wrap-around complexes with 1,3-dienes - ScienceDirect
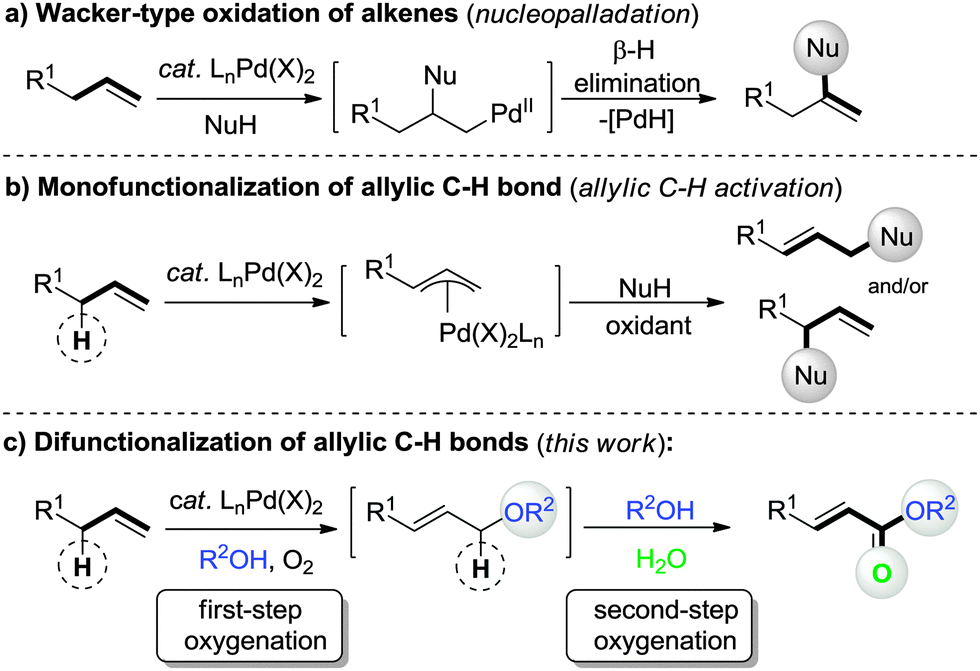
Palladium-catalyzed aerobic oxidative double allylic C–H oxygenation of alkenes: a novel and straightforward route to α,β-unsaturated esters - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC02277K

Integrating Allyl Electrophiles into Nickel‐Catalyzed Conjunctive Cross‐Coupling - Tran - 2020 - Angewandte Chemie International Edition - Wiley Online Library
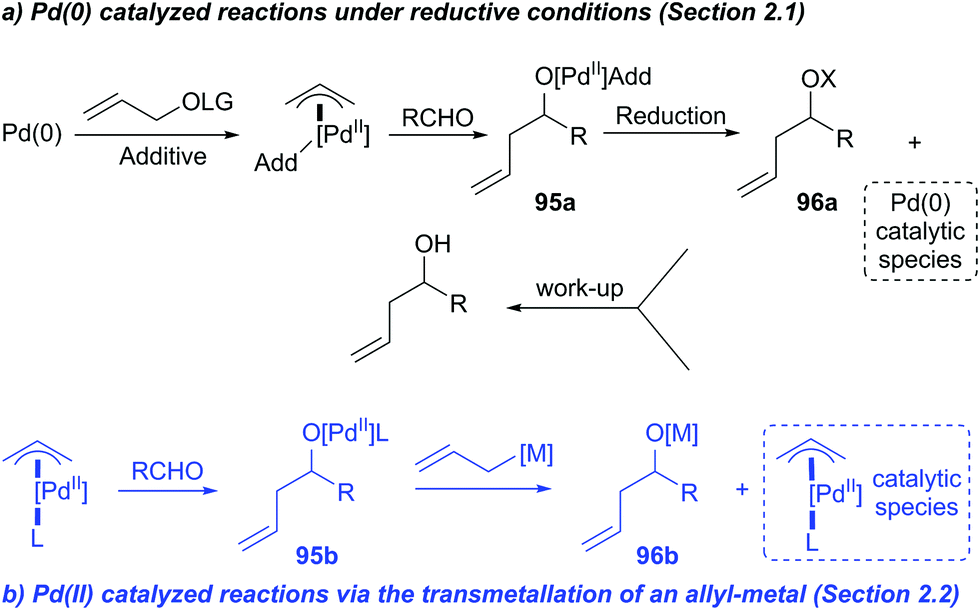
Catalytic nucleophilic 'umpoled' π-allyl reagents - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C7CS00449D

Nickel-catalyzed allylic carbonylative coupling of alkyl zinc reagents with tert-butyl isocyanide | Nature Communications
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01725A
![PDF] Synthesis of highly substituted pyrrolidines via palladium-catalyzed cyclization of 5-vinyloxazolidinones and activated alkenes | Semantic Scholar PDF] Synthesis of highly substituted pyrrolidines via palladium-catalyzed cyclization of 5-vinyloxazolidinones and activated alkenes | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/cc304cad8d2ab97404cd61ddbf74a80f56fbda8c/3-Figure2-1.png)
PDF] Synthesis of highly substituted pyrrolidines via palladium-catalyzed cyclization of 5-vinyloxazolidinones and activated alkenes | Semantic Scholar

Palladium-catalyzed enantioselective addition of two distinct nucleophiles across alkenes capable of quinone methide formation. | Semantic Scholar